Abstract
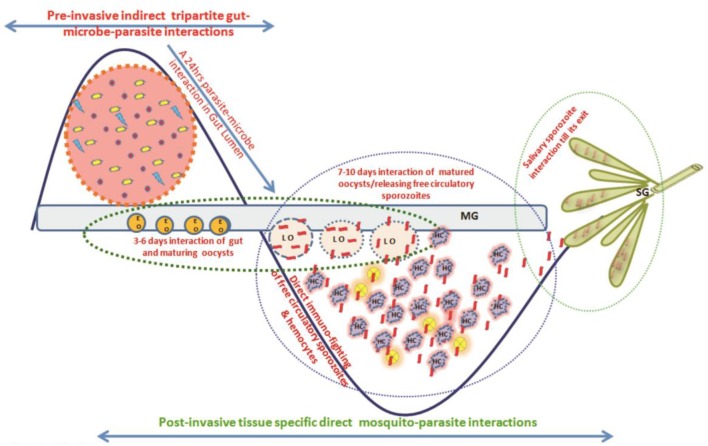
Blood-feeding enriched gut-microbiota boosts mosquitoes' anti-Plasmodium immunity. Here, we ask how Plasmodium vivax alters gut-microbiota, anti-Plasmodial immunity, and impacts tripartite Plasmodium-mosquito-microbiota interactions in the gut lumen. We used a metagenomics and RNAseq strategy to address these questions. In naïve mosquitoes, Elizabethkingia meningitis and Pseudomonas spp. are the dominant bacteria and blood-feeding leads to a heightened detection of Elizabethkingia, Pseudomonas and Serratia 16S rRNA. A parallel RNAseq analysis of blood-fed midguts also shows the presence of Elizabethkingia-related transcripts. After, P. vivax infected blood-meal, however, we do not detect bacterial 16S rRNA until circa 36 h. Intriguingly, the transcriptional expression of a selected array of antimicrobial arsenal cecropins 1-2, defensin-1, and gambicin remained low during the first 36 h-a time frame when ookinetes/early oocysts invaded the gut. We conclude during the preinvasive phase, P. vivax outcompetes midgut-microbiota. This microbial suppression likely negates the impact of mosquito immunity which in turn may enhance the survival of P. vivax. Detection of sequences matching to mosquito-associated Wolbachia opens a new inquiry for its exploration as an agent for “paratransgenesis-based” mosquito control.